A team including researchers from Khalifa University paves the way toward the design of new photocatalysts that can create useful products from CO2 using gold nanoparticles.
Excessive human-caused emissions of carbon dioxide, or CO2, have led to global warming. At the same time, however, CO2 is a nontoxic, inexpensive, abundant, and potentially reusable source of carbon. Converting it into high-value-added products such as plastics, paints, solvents, and fuel cells could be a viable and economic use of the carbon dioxide around us.
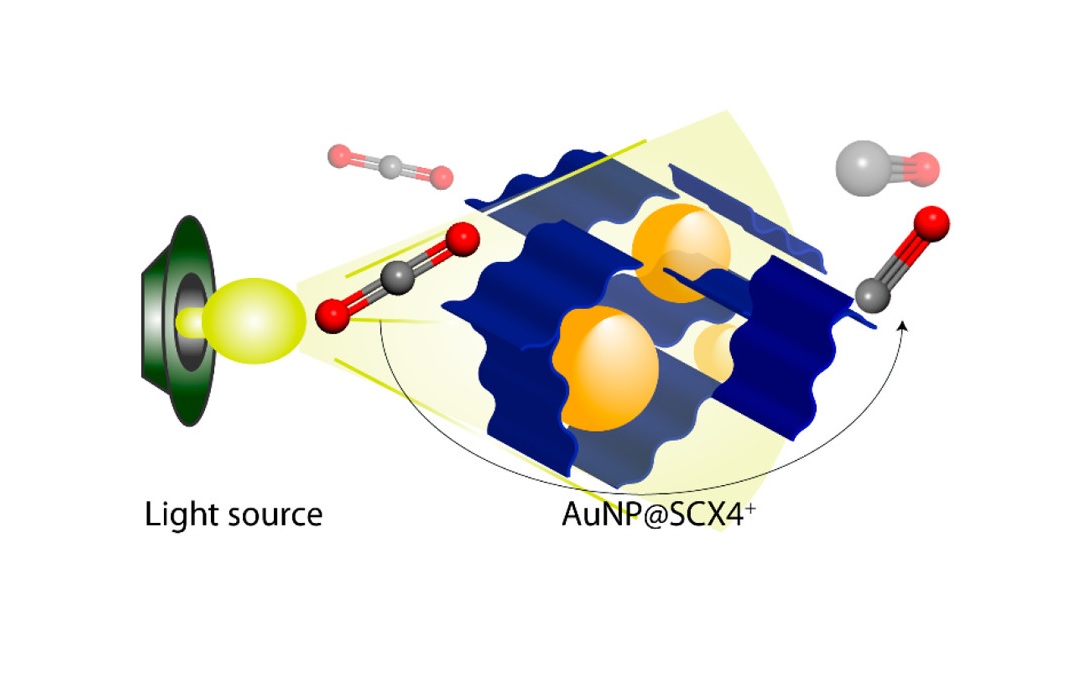
 A team of researchers from Khalifa University, New York University Abu Dhabi and the National Institute of Chemistry, Slovenia developed a novel structure, embedding gold nanoparticles in a polythiacalixarene to convert CO2 into viable products using natural light.
A team of researchers from Khalifa University, New York University Abu Dhabi and the National Institute of Chemistry, Slovenia developed a novel structure, embedding gold nanoparticles in a polythiacalixarene to convert CO2 into viable products using natural light.
The Khalifa University team, led by Dr. Dinesh Shetty, Assistant Professor of Chemistry, and a member of the Center for Catalysis and Separation (CeCaS), has developed an innovative material that could be useful to mitigate the negative environmental consequence of carbon emissions into the atmosphere. Their work was published in Applied Materials and Interfaces and accepted as a cover story.
Processes already exist to transform CO2 emissions into industrial chemicals although photocatalytic reduction of CO2 has been noted as particularly promising for the future. There’s little wonder why: it is based on photosynthesis. Green plants convert carbon dioxide and water into carbohydrates under ambient conditions using just sunlight, which is an inexhaustible and environmentally friendly energy source. Even better, photocatalytic CO2 reduction doesn’t create any secondary pollution.
“With the goal of producing a stable, durable, and recyclable heterogeneous catalyst that does not self-aggregate, several classes of materials have served as solid supports for metal nanoparticles,” Dr. Shetty explained. “Unfortunately, most of these supports have serious drawbacks. Activated carbons lead to leaching and aggregation at high temperatures. Metal-organic frameworks (MOFs) contain metal-ligand bonds that limit practical applications. Zeolites have very small pores that can effectively control metal nanoparticle growth but severely limit reactant access. In contrast, porous organic polymers (POPs) are chemically robust, have low skeletal density and a large pore volume that facilitates mass transfer of the reactants. Most importantly, POPs can be tuned to have sites that form strong bonds with the nanoparticles and ensure uniform distribution throughout the solid structure.”
The research teams decided to investigate gold nanoparticles as they are efficient absorbers of light and have been used for photocatalytic CO2 reduction in combination with other materials.
“Since light harvesting is the crucial first step of photocatalysis, we were interested in exploring the properties of gold nanoparticles in conjunction with a porous polymer,” Dr. Shetty said. The team synthesized a porous thiacalix[4]arene polymer and used it to anchor gold nanoparticles.
Calixarenes are bowl-shaped organic molecules that consist of defined hydrophobic cavities. This unique feature allows host-guest chemistry where calixarenes play the host role for small molecules and/or ions. Changing the surface chemistry enables Calixarenes to easily take on new functions, features, capabilities and properties. This is particularly true of calix[4]arenes, which contain four aromatic rings.
Research on metals on porous polymer supports has noted their high catalytic efficiency and the fact that the sulfur bridges in the calixarenes serve as nuclei for the growth of the metal nanoparticles. The research team’s material can be easily recycled and shows performance comparable to that of other materials, demonstrating the potential of gold nanoparticle porous polymer systems for photocatalytic applications.
Jade Sterling
Science Writer
29 August 2022