Center of Catalysis and Separation (CeCaS) scientific team recently co-authored/co-led a research paper in the prestigious journal Applied Catalysis B: Environmental, with an impact factor of 16.683 (Elsevier, Q1). The team is composed of G. Siakavelas (Visiting Scholar in CeCaS from University of Western Macedonia (UWM), Greece), Prof. M. Goula (UWM) and the KU PhD students Ayesha A. AlKhoori and Sara I. AlKhoori supervised by Prof. K. Polychronopoulou, Director of CeCaS.
The publication under the title “Highly selective and stable nickel catalysts supported on ceria promoted with Sm2O3, Pr2O3 and MgO for the CO2 methanation” came as a result of a fruitful collaboration with Prof. Maria Goula’s group from the UWM (CeCaS external partner). The doctoral student, G. Siakavelas, co-advised by both Prof.M. Goula and K. Polychronopoulou, kicked off this study while spending a visiting scholarship period in CeCaS research laboratories in 2018. During his stay he received training on the state-of-the-art synthesis and characterization methods by KU and CeCaS research staff and students. This study was further advanced from both sides, UWM and KU, after his departure from KU leading to a breakthrough on catalytic materials for the CO2 hydrogenation reaction.
In particular, this research explores the efficacy of the catalysts to convert the greenhouse gas carbon dioxide (CO2) into useful fuel, such as methane (Sabatier reaction) and highlights how the intrinsic properties of the catalysts can be tuned by using non-conventional methods of preparation and selected catalyst’s promoters. The significant role of this reaction can be understood based on the wide spectrum of methane applications; synthetic gas in the residential sector, raw material in different industries, and fuel for transportation. In addition, the CO2 to CH4 conversion is an important reaction for future manned space exploration to the red planet. The reaction is meant to utilize the Martian CO2-rich atmosphere to generate CH4 as propellant fuel and H2O as life support consumable. This research is ongoing and more exciting news are on the way.
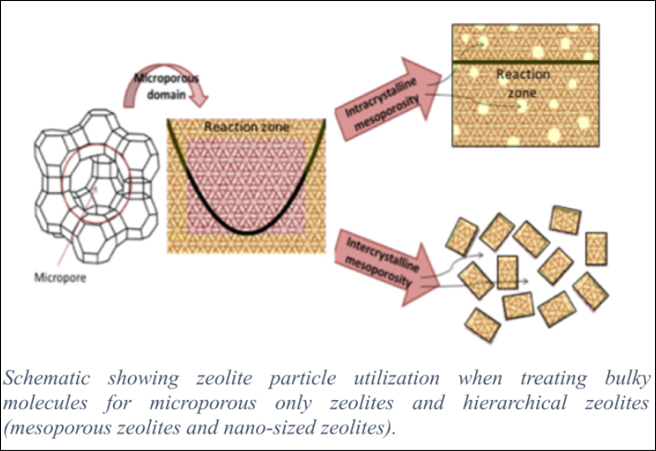
Even though fluid catalytic cracking (FCC) process started ~75 years ago, it is still the prominent conversion technology in the oil refinery to produce gasoline, propylene and other valuable products. In spite of efforts to find alternative energy resources to meet sustainability and environmental protection, the difficulties on massive production of renewable energy suggest continuing use of oil for many decades due to its maturity, efficiency and the availability of technology know-how. Undoubtedly, zeolites are vital catalysts and the major component in the FCC process to date. ~ 2,300 metric tons of FCC catalysts are produced per day, which means that ~0.16 kg of FCC catalysts are required for converting one barrel of feedstock.
However, the amount of utilization of zeolite catalysts depends on the feedstock. For instance, resid feedstock needs more catalyst than lighter feeds. Irrespective of the feed properties in FCC, occurrence of catalytic activity loss necessitates addition of certain percentage of fresh catalysts to the catalytic reactor on a daily basis in order to maintain the desired conversion. Therefore, increasing the stability of the zeolite phase can enhance the economics of the process. The recent addition of RFCC (residue FCC) in order to process heavy feed in refinery operations worldwide motivated researchers in both academia and industry to further explore ways to improve catalyst stability, activity and selectivity. We propose strategies to improve catalysts for RFCC applications by synthesizing zeolite Y with tunable (hierarchical) porosity.
Ayesha Abdulla Alkhoori is a PhD student in Prof. Kyriaki Polychronopoulou’s group. She works as a full-time student under the Center of Catalysis and Separation (CeCaS). Alkhoori is majoring in Materials Science and Engineering. Her PhD thesis is focused on developing materials for hydrocarbon
transformations coupling catalysis and separation. Ayesha gave two oral presentations in the 3rd ANQUE-ICCE International Congress of Chemical Engineering, which took place in Santander (Spain) from June 19th to 21st, 2019. The first talk titled “Improving Metal Oxide Catalysts for Biogas Dry Reforming: Coupling of Mechanochemical Modification with Enhanced Microwave Chemistry”. This research investigated the microstructure tailoring of ceria-based catalysts for dry reforming of methane reaction. Where the second presentation shed light on copper-ceria nanomaterials as catalysts for low temperature hydrogen purification and carbon monoxide capturing in proton exchange membrane fuel cells (PEMFC).
This international congress is a reference for researchers in the field of chemical engineering and chemistry applied to the industry. Alkhoori had the opportunity to exchange up-to-date information and connect with researchers and experts from different universities and countries.
Sara Alkhoori is a full-time MSc student in mechanical engineering under the supervision of Assoc. Prof. Kyriaki Polychronopoulou. Her MSc thesis is focused on improving metal oxide catalysts for biogas dry reforming reaction by coupling mechanochemical synthesis with enhanced microwave chemistry. Earlier this June, she was invited to visit university of Surrey in England during the period 15th to 30th of June for an organized study program on surface analysis training using the state-of-art x-ray photoelectron spectroscopy (XPS).
This technique is used to analyze the surface chemistry of a material by identifying the elements on the surface and quantifying their elemental compositions and chemical environment. Therefore, the use of it in catalysis is expected to deepen the overall understanding of the chemical behavior of a catalytic surface. Sara had a great opportunity to visit the Surface Analysis Laboratory at Surrey and engage with the experts in surface science, Dr. Mark Baker (Reader) and Dr Steve Hinder (Research scientist). Being trained on XPS will further her academic studies as a postgraduate research student at Khalifa university and enrich her knowledge in surface sciences.

Mrs. Margarida Ferreira, was a visiting student of the Center of Catalysis and Separations (CeCaS) supporting the modeling work we are doing in CeCaS under Theme 3 ‘Multi-scale Modeling’, and embracing the existing collaboration between the research group she belongs to in Lisbon (Portugal) and our KU research group. Mrs. Margarita Ferreira, presented the joined work in the International Conference on Properties and Phase Equilibria for Product and Process Design, PPEPPD, held in Vancouver, CANADA, on May 12-16, 2019. [Photo: Mrs. Ferreira explaining her poster to Prof. Joan Brennecke, the editor in chief of the Journal of Chemical and Engineering Data (American Chemical Society)]
Two PhD students under the supervision of Dr. Kyriaki Polychronopoulou, Associate Professor of Mechanical Engineering and Director of the KU Center for Catalysis and Separation (CeCaS), presented papers at the Sustainable Industrial Processing Summit & Exhibition (SIPS), which took place in Cyprus in October, demonstrating KU’s strong research capabilities in the field of catalysis.
Ayesha Alkhoori presented a paper titled “H2 Production from Glycerol Steam Reforming.” Her paper describes how she has developed an improved catalyst capable of producing high yields of hydrogen by steam reforming glycerol, a by-product of biodiesel production, at a low temperature.
A recent spike in the production of biodiesel in the UAE – brought about by local efforts to reduce the country’s carbon footprint through greener fuels – has led to an increase in the production of crude glycerol. “For every one ton of biodiesel, 100 kg of crude glycerol is generated as the main byproduct,” Alkhoori explained.
While glycerol is an important feedstock for the manufacture of value-added chemicals and clean fuels, including hydrogen, most of this surplus crude glycerol gets incinerated. This is because current methods for turning glycerol into useful chemicals – which is done through a catalytic process called glycerol steam reforming – requires very high operating temperatures above 500 degrees Celsius, and high costs.
Currently, when glycerol is steam reformed using a nickel-based catalyst, carbonaceous chunks of waste called ‘coke’ build up on the surface of the catalyst, blocking the reaction sites and reducing efficiency. To overcome this issue, Alkhoori designed a catalyst made of copper and cerium to be highly reducible, which means that it provides the key ingredient of oxygen to the reaction sites, to convert the glycerol into hydrogen gas more efficiently. The activated oxygen carries out the chemical reactions at lower temperatures and lower cost, without sacrificing the hydrogen yield.
The research began last year at CeCaS under the supervision of Dr. Kyriaki Polychronopoulou, Associate Professor of Mechanical Engineering, and involves collaborators from the University of Western Macedonia. This work was published in early 2019 in the Sustainable Energy & Fuels, journal of the Royal Society of Chemistry (UK).
A second PhD student, Aseel Hussein, presented a paper titled “Improving Metal Oxide Catalysts for Biogas Dry Reforming: Coupling of Mechanochemical Modification with Enhanced Microwave Chemistry.” Her paper, which was done in collaboration with MSc student Sara AlKhoori, describes a ‘smart’ design for a catalyst used to convert methane, a greenhouse gas, into a combination of carbon monoxide and hydrogen known as syngas, which can be used to produce ethanol, methanol, or other clean liquid fuels.
Methane is converted into syngas through a process known as “dry reforming of methane.” The process is typically carried out with noble metal elements, which are known for their high activity and stability. But noble metal elements are very costly, making the process too expensive to perform at an industrial scale.
Hussein proposes using catalysts made from much lower cost nickel and cobalt. The challenge with these type of transition-based catalysts, however, is that they are prone to coking, which blocks the reaction sites and ultimately stops the catalytic activity.
To get around this issue, Hussein designed her nickel-based catalysts with increased oxygen storage capacity. By increasing its oxygen storage capacity, the catalyst can leverage the carbon buildup for more efficient gasification, which is process needed to convert the methane into syngas.
Hussein’s research started in 2017 and is being sponsored by the Abu Dhabi Department of Educational and Knowledge (ADEK) through the Research Excellence Award (AARE) 2017. She is also being supervised by Dr. Polychronopoulou. She is collaborating with research teams from University of Western Macedonia in Greece, the University of Surrey in the UK, and University of Zaragosa in Spain. She plans to continue testing the best catalytic system in terms of selectivity and stability to better control the coking.