A simple and straightforward method can be used to develop catalysts for the chemical process that turns carbon dioxide emissions into high-energy-density fuels. Nickel nanoparticles and cerium(IV) oxide combine to make a catalyst more than 50 percent more effective than already existing catalysts.
A team of researchers including Dr. Aasif Dabbawala, Prof. Kyriaki Polychronpoulou and Aseel Hussein from Khalifa University’s Center for Catalysis and Separations (CeCaS) has developed a new catalyst for transforming carbon dioxide emissions into fuels that could be used for space travel and Mars exploration missions.
The CeCaS team collaborated with researchers from University College London, Cyprus University of Technology, the University of Surrey, University of Western Macedonia, Greece, and Universidad de Zaragoza, Spain. Their results were published in Applied Catalysis B: Environmental.
“Following the industrial revolution, the sharp increase in anthropogenic carbon dioxide emissions has caused a great spike in its concentration in the atmosphere,” Prof. Polychronopoulou said. “Capturing and converting part of this emitted CO2 towards value-added products aims to mitigate the adverse effects of climate change, while transitioning towards a net-zero economy. CeCaS has a very diverse portfolio of technologies under development for decarbonization.”
One such value-added product is the development of high-energy-density fuels such as methane and jet fuel. Captured carbon dioxide can be converted via hydrogenation into these fuels, circumventing problems related with the storage and transportation of hydrogen, since hydrogen has a very low volumetric energy density. Transforming CO2 into methane (methanation) has also been indicated as potentially useful for Mars exploration missions, since abundant CO2 from the Martian atmosphere could be used to generate fuel for methane-powered rocket engines.
The catalysts used to power this methanation reaction typically use nickel, ruthenium, or rhodium, but nickel-based catalysts are much more likely to be used due to their high activity and lower cost. Beyond the material used, however, the structure of the catalyst itself is vital in producing the most effective catalyst for methanation. The support structure can provide the active sites for the adsorption and activation of the carbon dioxide and enhance the dispersion of the nickel nanoparticles.
Cerium(IV) oxide (CeO2) is a metal oxide often used as an oxidant and catalyst in chemical processes.
“Nickel-based CeO2 catalysts are reported in numerous studies to be greatly superior to nickel catalysts supported on other metal oxides, especially in relation to low-temperature activity and methane selectivity,” Prof. Polychronopoulou said.
The chemical bond between the oxygen and the cerium is easily broken, which promotes the adsorption of the carbon dioxide into the active sites and enhances the conversion and removal of the reaction intermediates. This makes the methanation process more efficient.
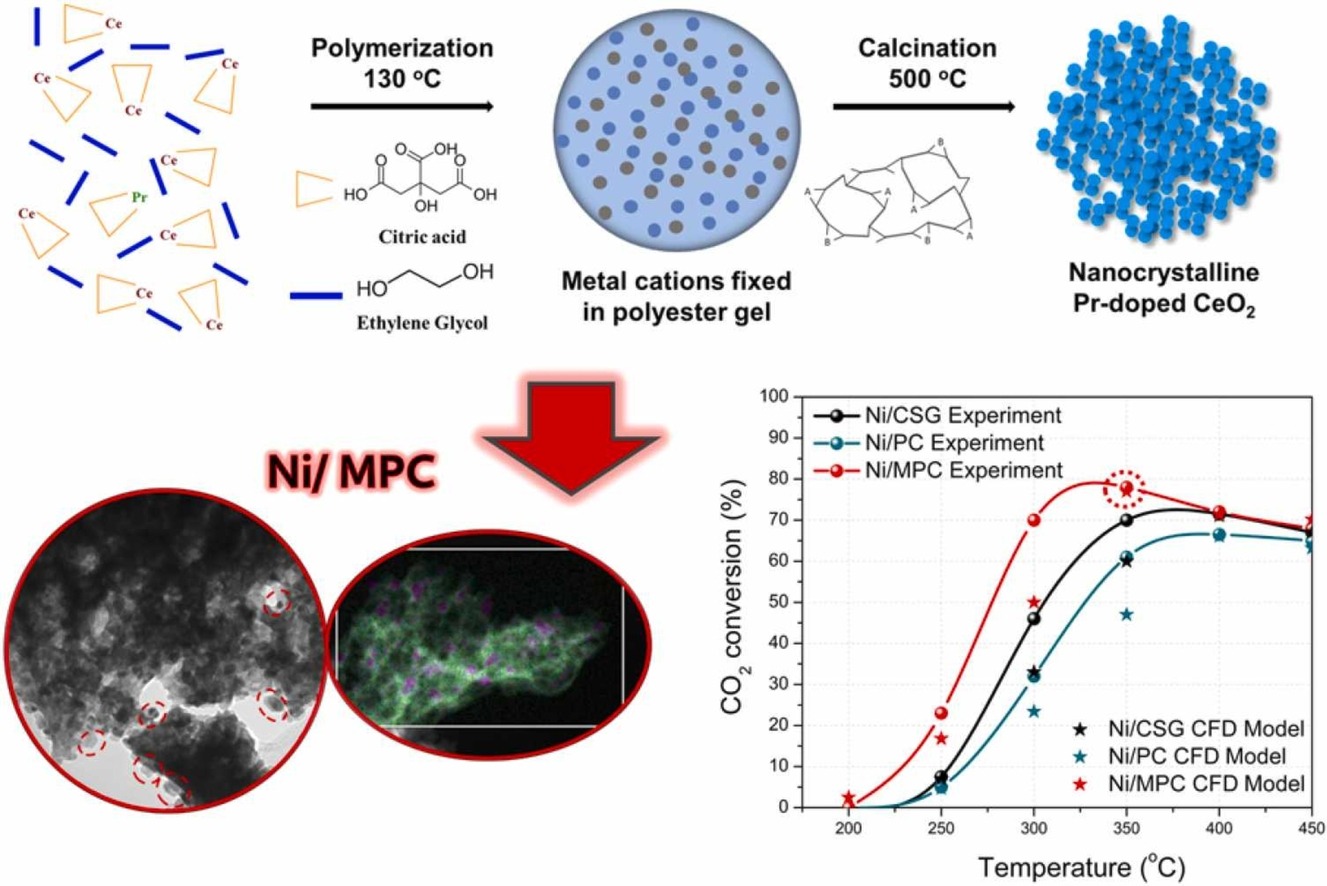
Adding small amounts of praseodymium (Pr) to the nickel-based CeO2 catalyst can also enhance the methanation process. The research team developed their catalyst using Pr-doped CeO2 and nickel nanoparticles. A wide spread of the nickel nanoparticles translates to a more efficient catalyst, which the research team achieved using a modified synthesis technique.
“Another important factor that needs consideration is the architecture or nanostructure of the CeO2-based support,” Prof. Polychronopoulou added. “Surface defect chemistry and the available surface area are just as important as the nickel dispersion and mean size of the supported nickel nanoparticles in affecting the catalytic activity.”
An effective catalyst needs lots of activation sites — pores in the structure where the carbon dioxide can collect and adsorb for the methanation process to occur. The team used a modified sol-gel-type synthesis process to create the porous architecture in the Pr-doped CeO2 supports. The sol-gel process is a method for producing solid materials from small molecules, used for the fabrication of metal oxides. In this case, the CeO2. They then deposited the nickel nanoparticles onto the supports.
“Using ethylene glycol in the absence of water during our modified synthesis process led to a metal oxide support with larger pore size and volume, which was conducive to the deposition of medium-sized nickel nanoparticles into the nanoporous structure,” Prof. Polychronopoulou said. “The high nickel dispersion and availability of surface defects and basic sites greatly improved the catalyst’s activity.”
The research team saw a 50 percent increase in carbon dioxide conversion at 300C, compared with other catalysts. They conclude that the catalyst nanostructure and active metal dispersion play a crucial role toward the design of carbon dioxide methanation catalysts. Theirs is a simple and straightforward method to produce metal oxide supports with tailored nanopore structures, which allows the production of highly active catalysts.
Jade Sterling
Science Writer
15 November 2022