Two patients with unique genetic mutations in a single gene sparked the investigation of 40 researchers into the effects of gene expression on diabetes
The discovery and mapping of the complete human genome in 2003 introduced the possibility of individualized medicine to a person’s physical and genetic makeup. Increasing evidence is now demonstrating that a patient’s unique genetic profile can be used to detect a disease’s onset, prevent its progression, and optimize its treatment.
This has led to enhanced global efforts to implement precision (personalized) medicine and pharmacogenomics in clinical practice. One such area of clinical practice is the treatment of diabetes.
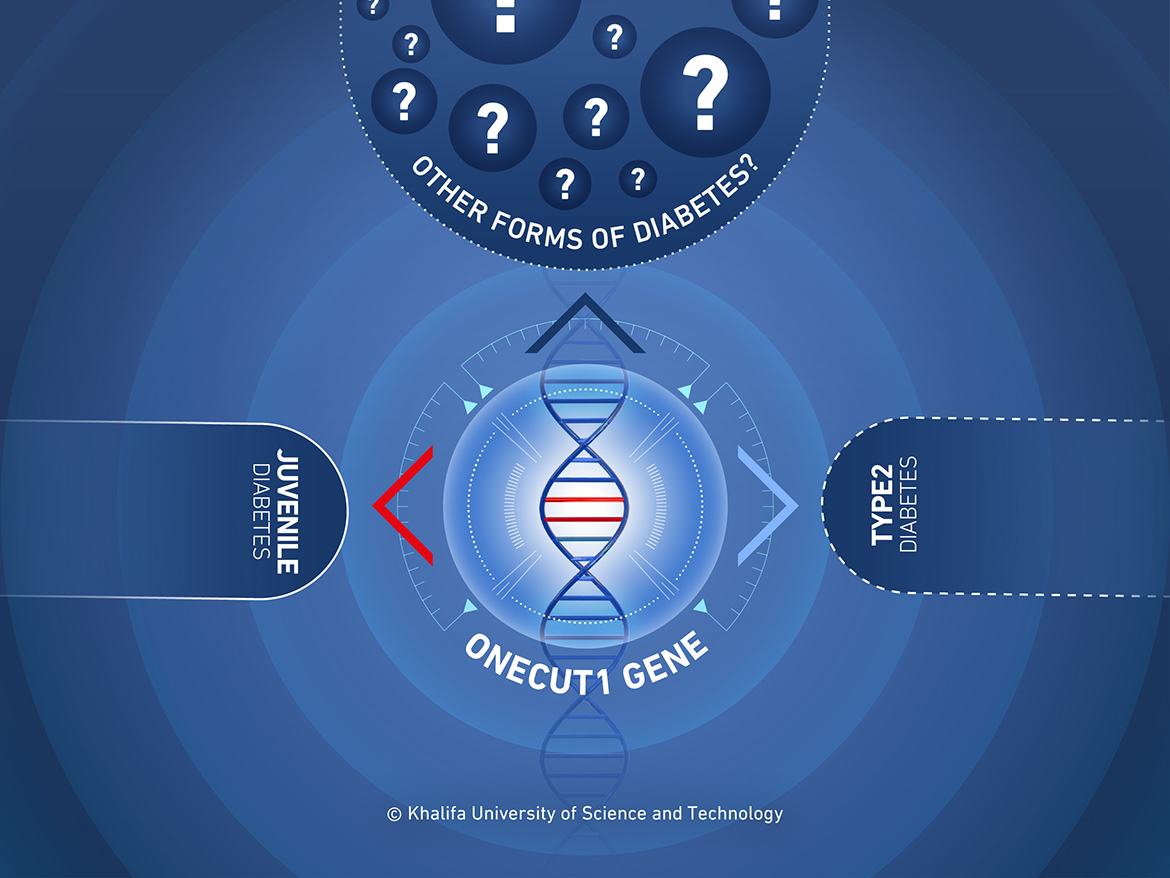
A team of 40 researchers from around the world has studied the genomes of patients with diabetes and their families to identify one of the genes responsible for monogenic diabetes—diabetes caused by mutations in a single gene. In contrast, the most common types of diabetes are caused by multiple genes or lifestyle factors. Most cases of monogenic diabetes are inherited.
Dr. Pierre Zalloua, Professor and Chair of the Department of Molecular Biology and Genetics, collaborated with researchers from France, Germany, Austria, the United States, and Singapore to determine the gene responsible for two cases of monogenic diabetes. Their results were published in Nature Medicine.
“Diabetes affects over 350 million people worldwide, and the discovery and study of genes responsible provide important insights for understanding disease mechanisms,” Dr. Zalloua explained. “With better understanding, we can improve quality of life and develop cost-effective care for diabetes patients.”
Diabetes mellitus is a group of metabolic diseases, all of which are characterized by high blood glucose levels. If left untreated, diabetes can lead to severe complications including blindness, kidney and heart disease, stroke, loss of limbs, and reduced life expectancy. It is a major public health problem, affecting hundreds of millions of people worldwide and representing a substantial economic burden on society.
There are two types of diabetes: Type 1 and Type 2 diabetes. Type 1 usually begins in childhood with individuals suffering from their body’s inability to produce enough insulin, while Type 2 is commonly associated with obesity and usually occurs during middle age. Both types tend to run in families and genetic factors contribute to the disease, with interactions between genetic and environmental factors being critical.
“Several genes are shared between monogenic diabetes and multifactorial Type 2 diabetes, suggesting that shared disease mechanisms may exist,” Dr. Zalloua said. “Remarkably, many of these genes encode key proteins for pancreas development.”
To determine which genes play a part in the development of diabetes, the research team examined two different patients with diabetes: one, a young French boy with neonatal diabetes, and a second Turkish child with diabetes diagnosed at 14 months. They showed that the patients inherited mutated alleles of one particular gene, ONECUT1. Two mutated alleles led to a severe form of neonatal diabetes where the child developed a small pancreas and a missing gall bladder, while one mutated allele saw an increased risk of diabetes in the second patient. The researchers were able to determine that ONECUT1 and its expression is a major player in diabetes.
Dr. Zalloua was the person who originally identified additional cases from the region linked to this gene, including a case from a patient in Lebanon. Analysis of these patients revealed various different ONECUT1 mutations, all linked to a risk of diabetes.
ONECUT1 affects a variety of processes including glucose metabolism, an important factor in the disease mechanism of diabetes. Its expression also influences the development of the pancreas and the gallbladder. Previous studies of ONECUT1 have focused on the gene’s role in retinal development, but it is now clear that ONECUT1 acts to determine what type of cell a stem cell becomes. Some human stem cells are pluripotent, meaning they can become any kind of cell in the body, and genes including ONECUT1 are the deciders. Mutations in this gene can therefore disrupt a very complex process at various stages.
The pancreas plays an essential role in converting food to fuel in the body: it helps in digestion and in regulating blood sugar. Two of the main pancreatic hormones are insulin, which acts to lower blood sugar, and glucagon, which acts to raise blood sugar. A functioning healthy pancreas automatically produces the right amount of insulin; in people with diabetes, the pancreas either produces little or no insulin, or the cells do not respond to the insulin that is produced.
To further validate their findings, the researchers examined a cohort of over 2000 German people with presumed type 2 diabetes, and identified 13 incidences of ONECUT1 mutations. In another, larger and multi-ethnic, cohort of almost 20,000 people with type 2 diabetes, the researchers also found that people with variants of the ONECUT1 gene were more likely to develop type 2 diabetes. However, they noted that the risk varied with the specific variant.
While monogenic cases of diabetes may be rare, and monogenic cases of ONECUT1-diabetes even rarer, this form of diabetes is also the most suited to a precision medicine approach. Identifying the cause means we can pinpoint the best treatment, offering an opportunity to shift focus from broad population-based standards of care to tailored treatments targeted to an individual molecular profile.
“We found that ONECUT1 controls mechanisms regulating endocrine development, which is involved in a wide spectrum of diabetes types,” Dr. Zalloua said. “We highlighted the broad contribution of ONECUT1 to diabetes pathogenesis, marking an important step towards precision medicine for diabetes.”
Jade Sterling
Science Writer
18 January 2022